Adavosertib and beyond: Biomarkers, drug combination and toxicity of WEE1 inhibitors - ScienceDirect

Ado-trastuzumab emtansine (T-DM1) in patients with HER2-amplified tumors excluding breast and gastric/gastroesophageal junction (GEJ) adenocarcinomas: results from the NCI-MATCH trial (EAY131) subprotocol Q - ScienceDirect

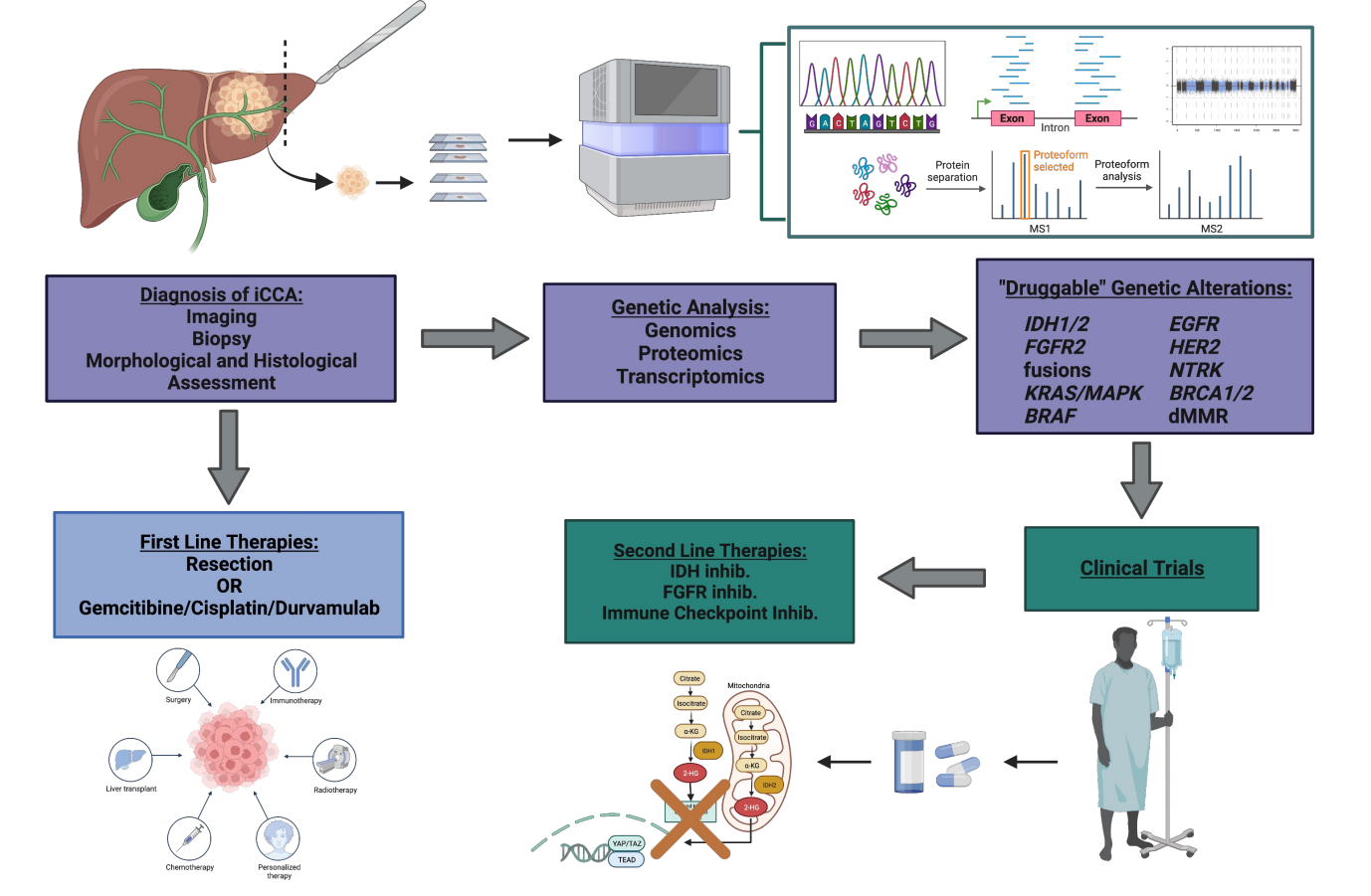
Full article: Druggable molecular alterations in bile duct cancer: potential and current therapeutic applications in clinical trials

Adavosertib and beyond: Biomarkers, drug combination and toxicity of WEE1 inhibitors - ScienceDirect
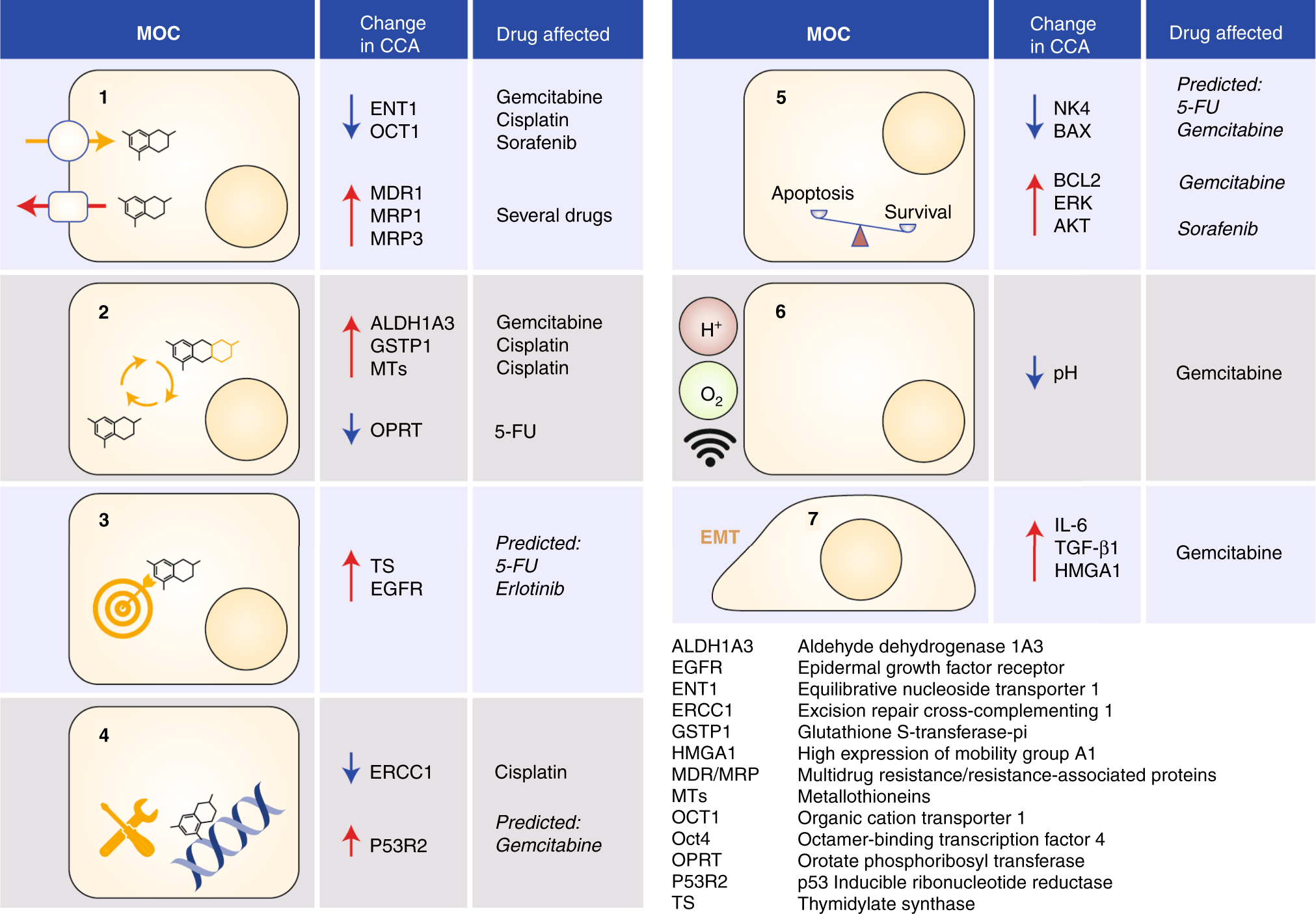
Current and novel therapeutic opportunities for systemic therapy in biliary cancer | British Journal of Cancer

Conceptual Framework for Therapeutic Development Beyond Anti–PD-1/PD-L1 in Urothelial Cancer | American Society of Clinical Oncology Educational Book
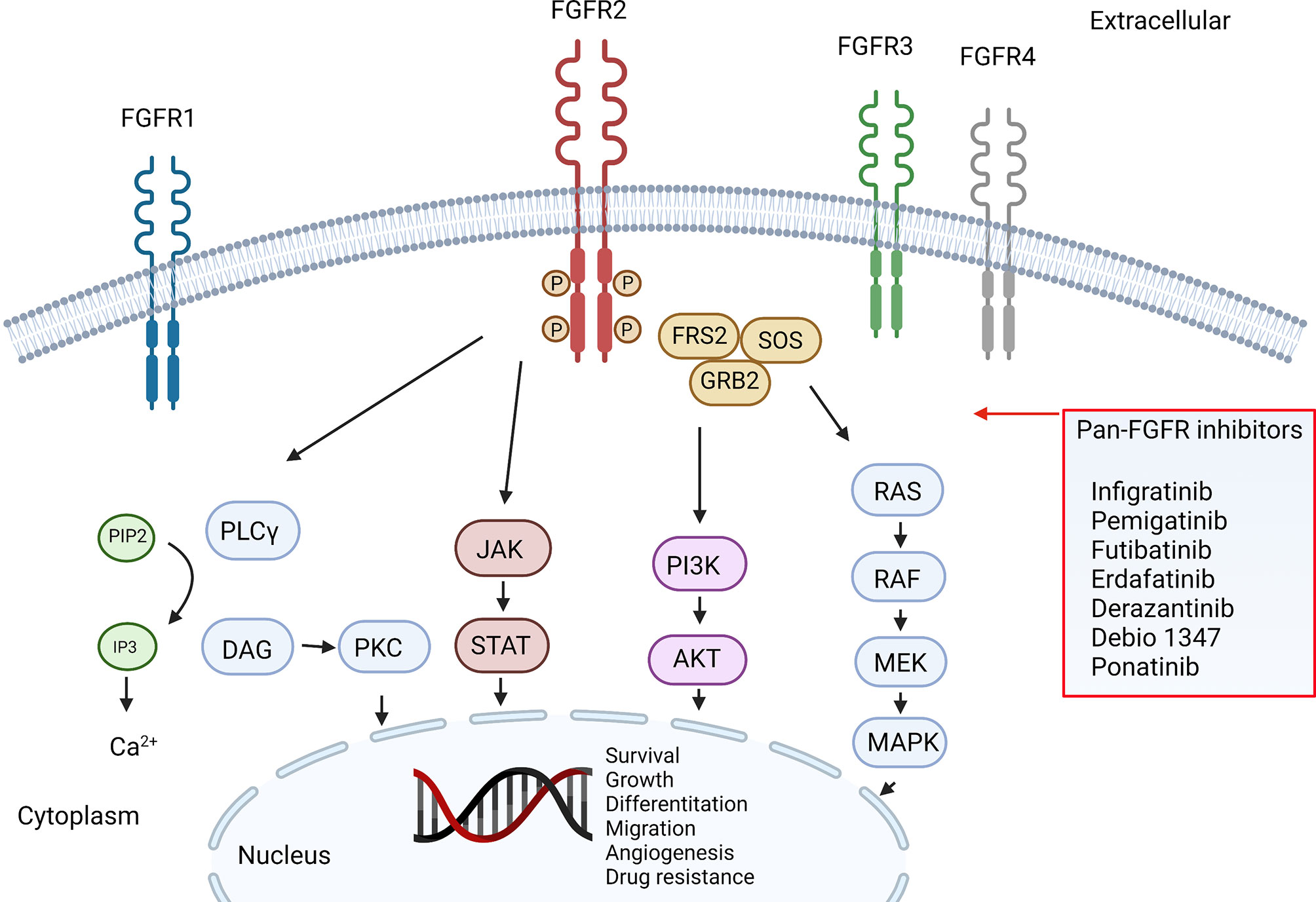
Frontiers | Precision Medicine Targeting FGFR2 Genomic Alterations in Advanced Cholangiocarcinoma: Current State and Future Perspectives

Overall survival with sacituzumab govitecan in hormone receptor-positive and human epidermal growth factor receptor 2-negative metastatic breast cancer (TROPiCS-02): a randomised, open-label, multicentre, phase 3 trial - The Lancet

Pertuzumab and trastuzumab for HER2-positive, metastatic biliary tract cancer (MyPathway): a multicentre, open-label, phase 2a, multiple basket study - The Lancet Oncology

Abbreviations: GEMCIS, Gemcitabine and cisplatin combination therapy;... | Download Scientific Diagram

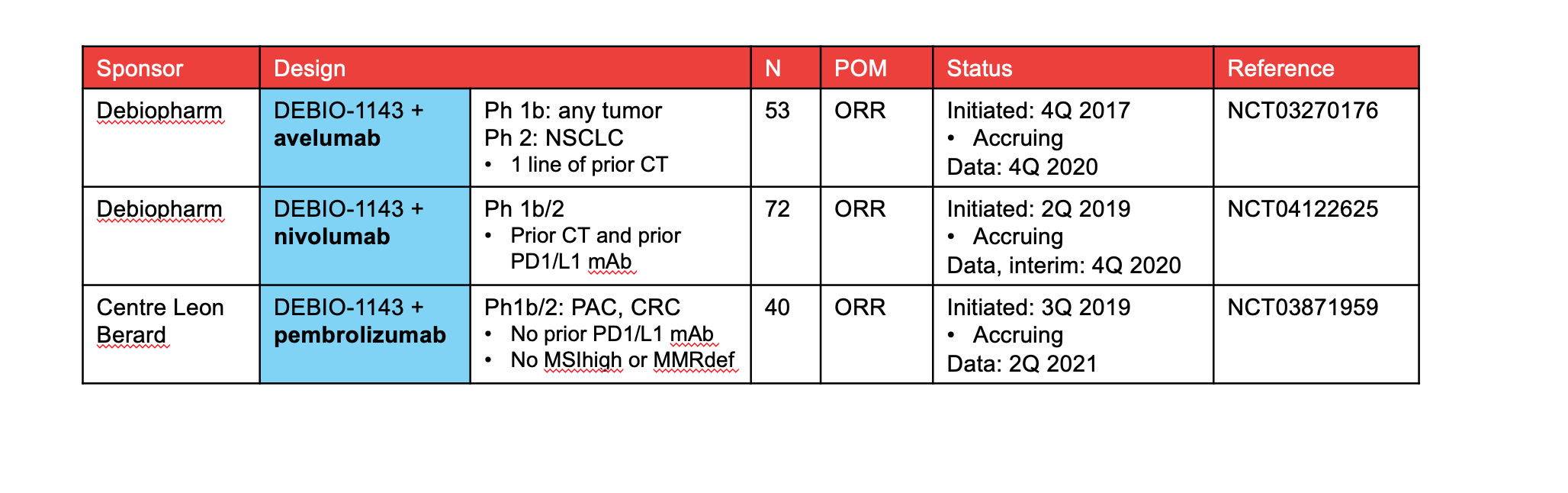
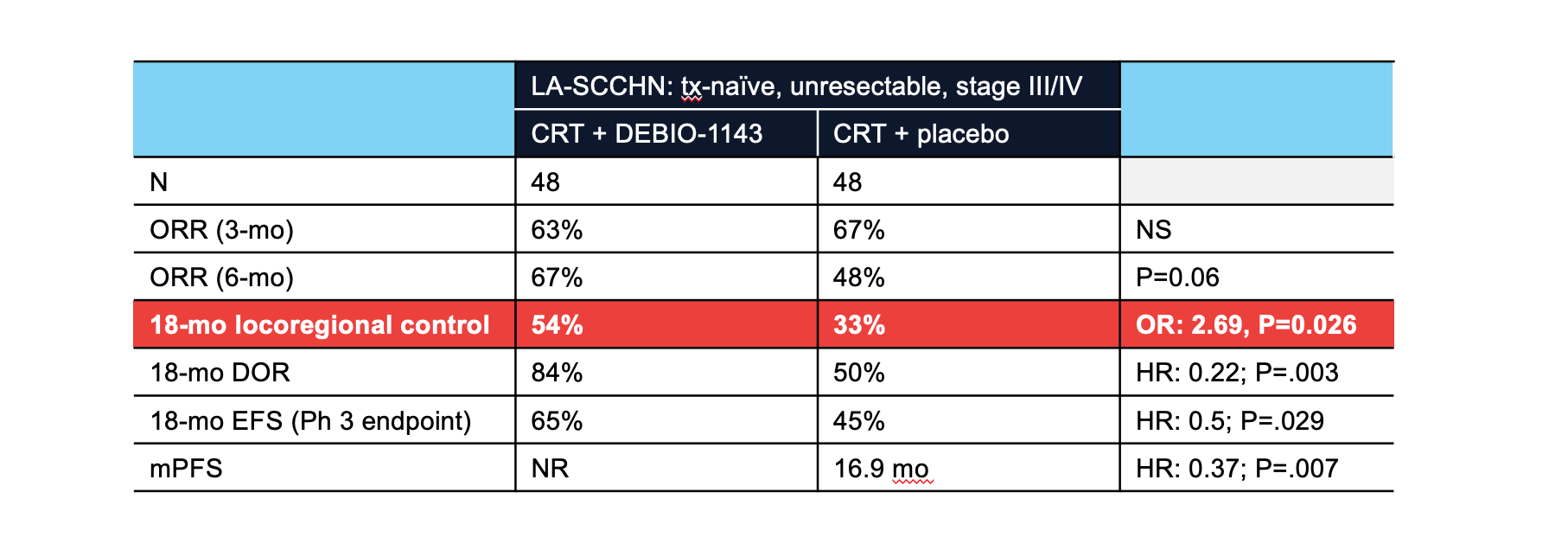
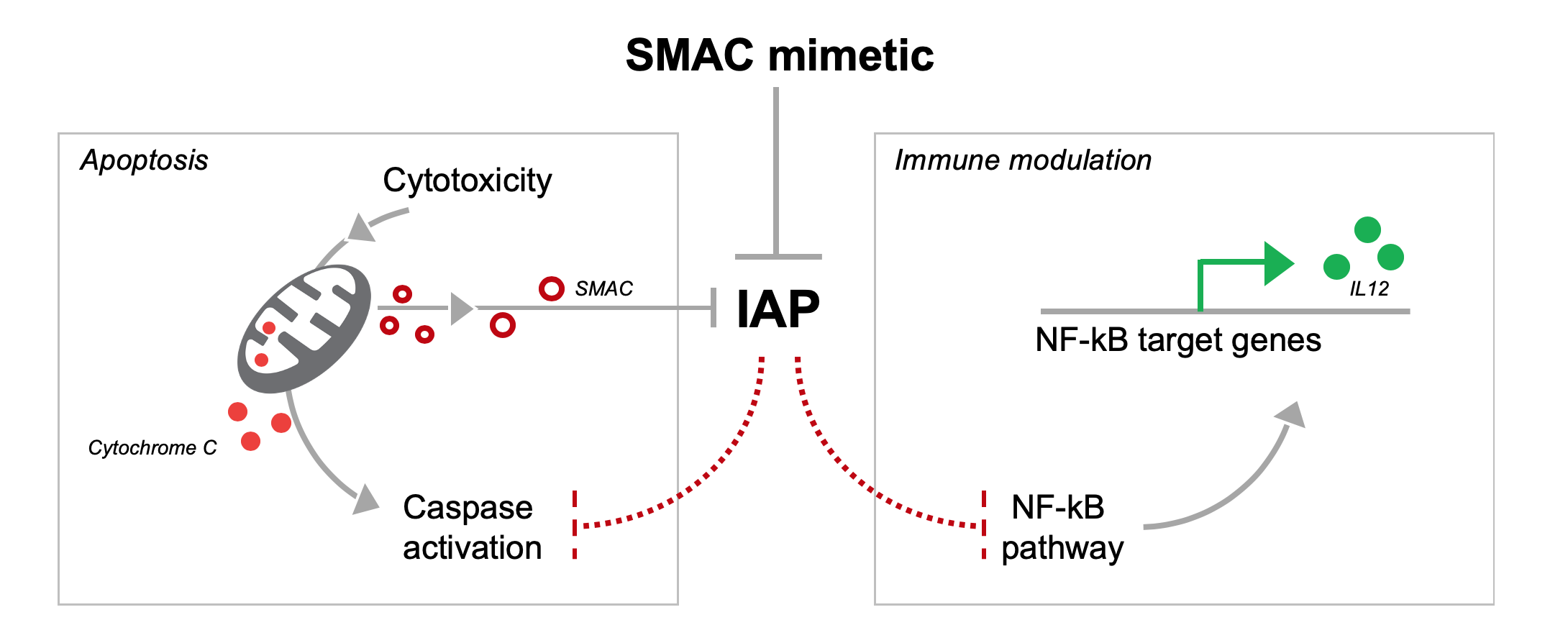
PDF) 560P Safety and efficacy of Debio 1143, an antagonist of inhibitor of apoptosis proteins (IAPs), in combination with nivolumab in a phase Ib/II trial in patients (pts) failing prior PD-1/PD-L1 treatment

Pertuzumab and trastuzumab for HER2-positive, metastatic biliary tract cancer (MyPathway): a multicentre, open-label, phase 2a, multiple basket study - The Lancet Oncology
Open-label, non-randomized, exploratory pre-operative window-of-opportunity trial to investigate the pharmacokinetics and pharma

Molecular targeted and systemic therapy for intrahepatic cholangiocarcinoma: a multi-disciplinary approach | Future Oncology

Ado-trastuzumab emtansine (T-DM1) in patients with HER2-amplified tumors excluding breast and gastric/gastroesophageal junction (GEJ) adenocarcinomas: results from the NCI-MATCH trial (EAY131) subprotocol Q - ScienceDirect
First-in-human Phase I “Basket” Study of Debio 1347 (CH5183284), a Novel FGFR Inhibitor in Patients with FGFR Genomically